| Company: | AcelRx Pharmaceuticals, Inc. |
| Ticker Symbol: | ACRX |
| Class Period: | Dec-2-13 to Sep-25-14 |
| Date Filed: | Oct-3-14 |
| Lead Plaintiff Deadline: | Dec-2-14 |
| Court: | Northern District of California |
| Allegations: | |
The lawsuit alleges that defendants made false and/or misleading statements concerning its Zalviso system pain management product. In particular, the complaint alleges that: (1) the Instructions for Use (IFU) for Zalviso were not designed to adequately address the risk of the accidental misplacement of tablets; (2) AcelRx had not submitted to the FDA sufficient data to support the shelf life of Zalviso; and (3) as a result of the foregoing, AcelRx's statements concerning Zalviso, including the drug's regulatory approval and financial prospects, were materially false and misleading.
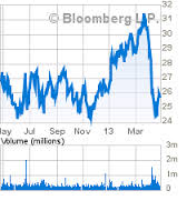
On July 25, 2014 AcelRx announced that it had received a Complete Response Letter ("CRL") from the FDA regarding its New Drug Application for Zalviso. The FDA requested additional information on the Zalviso System to ensure proper use of the device, including changes to the Instructions for Use for the device and additional data to support the shelf life of the product. This news caused shares of AcelRx to decline $4.44/share, or 40%, the next trading day.
Then when AcelRx disclosed on September 26, 2014 that the resubmission process for its Zalviso NDA would not be complete until the first quarter of 2015, and that the planned resubmission will have a review period of six months, shares of AcelRx declined $1.31/share, or over 19%, causing investors substantial losses.
If you acquired the securities of the defendants during the Class Period you may, no later than the Lead Plaintiff Deadline shown above, request that the Court appoint you as lead plaintiff through counsel of your choice. You may also choose to remain an absent class member. A lead plaintiff must meet certain requirements.