 According to an exhaustive investigation by the Washington Post (7/19/12), a significant body of research also suggests the anemia drugs carry potentially lethal side effects, such as cancer and strokes.
According to an exhaustive investigation by the Washington Post (7/19/12), a significant body of research also suggests the anemia drugs carry potentially lethal side effects, such as cancer and strokes.The Post highlighted the story of cancer patient Jim Lenox, who was administered a shot of anemia drugs as he was waiting to be discharged from the cancer clinic at the Baltimore Washington Medical Center. As reported by the Post, his wife felt it somewhat odd given her husband's blood readings had been close to normal (anemia drugs are often used for cancer and kidney dialysis patients to elevate a patient's red blood cell count).
Why did her husband need the expensive shot? And what role did a dose of medication worth over two thousand dollars have on a dying man's life? The Post reported that within hours of Lenox having been administered the shot, he was dead.
There is no question anemia drugs have performed well for their respective manufacturers. Amgen and Johnson & Johnson are reported to have made about $8 billion each, per year, from anemia drugs. Doctors have been keen to prescribe them, based on the promoted benefits.
But those benefits are now becoming suspect according to The Post's investigative report, citing long-standing agreements between the US Food and Drug Administration (FDA) and the manufacturers. What they found, it appears, is that anemia drugs with their so-called 'life benefits' were approved based on clinical trials that missed, or glossed over the risks while highlighting the alleged benefits. More recent attempts to restrict large doses were met with an intense lobbying effort on behalf of the manufacturers. Congress' hand was forced into allowing the largest doses of anemia drugs to retain FDA approval.
The emerging result is that anemia drugs such as Epogen, Procrit and Aranesp have cost private and public health plans such as Medicaid and Medicare billions of dollars in allegedly unnecessary costs. Many states, if history is a guide, might consider launching lawsuits in an effort to try and get some of those dollars back.
Medication that is billed at $2,500 per dose, will add up quickly.
Meanwhile, a whistleblower lawsuit involving anemia drugs fed to kidney dialysis patients has resulted in a $55 million settlement over allegations of drug overuse.
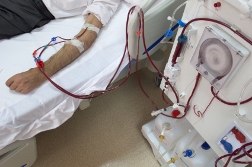
DaVita Inc., based in Denver and a force in the kidney dialysis industry, admitted no wrongdoing in the lawsuit. The issue was DaVita's use of Epogen, the anemia drug made by Amgen. According to the Denver Post (7/4/12), kidney dialysis patients requiring the involved procedure upwards of three times per week, can easily find themselves anemic. Epogen, therefore, is commonly used to restore dialysis patient's red blood cell counts to healthy levels.
In DaVita's case, the whistleblower lawsuit was based on an accusation that DaVita used more Epogen than was medically necessary, and for double-billing the US government for Epogen left over in vials and reused.
"DaVita and its affiliated physicians did nothing wrong and stand by their anemia management practices, which were always consistent with their mission of providing the best possible care for each patient," a company statement said.
However, amidst recent revelations concerning the true benefits of anemia drugs v. their risks and their enormous costs, the benefits to patients may be suspect. The benefits to the manufacturers—billions of dollars in revenue—are of little doubt.




